Ozempic oral tablet approved for type 2 diabetes in U.S., offering weekly injection alternative.
- FDA approves Ozempic oral tablet for type 2 diabetes
- Replaces Novo Nordisk’s daily Rybelsus pill
- Oral version offers smaller doses than injections
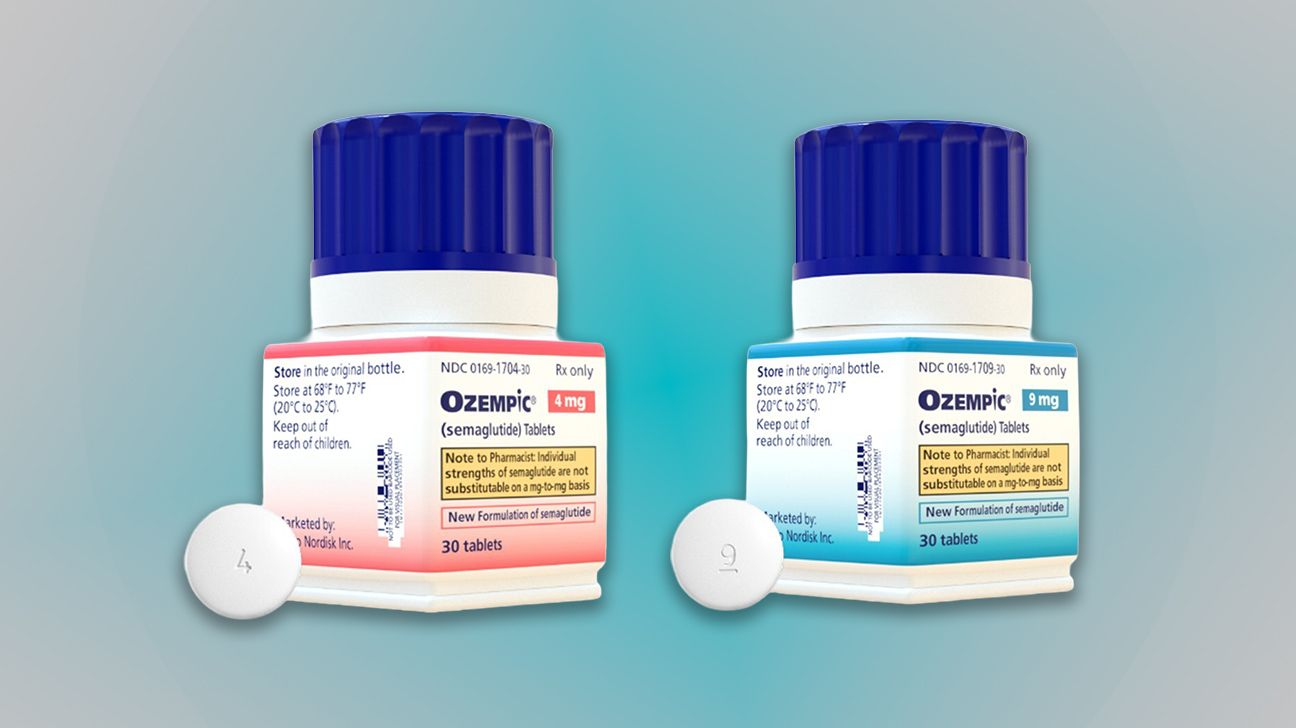
The U.S. Food and Drug Administration has approved a new oral tablet formulation of Ozempic, the widely prescribed GLP-1 medication for type 2 diabetes. Manufactured by Novo Nordisk, the oral tablet became available on May 4 for adults diagnosed with the condition. The drug replaces Rybelsus, Novo Nordisk’s previous daily oral medication approved in 2019 for type 2 diabetes.
Experts say the new Ozempic pill may offer a more convenient option for patients who avoid weekly injections. The oral tablet comes in smaller doses compared to the injectable version, which has become increasingly popular for its weight loss benefits alongside diabetes management. Clinical trials showed the pill maintained similar efficacy to the injectable form in controlling blood sugar levels.
Novo Nordisk reformulated Ozempic into an oral tablet to address patient preferences. The company discontinued Rybelsus on the same day the new pill launched, signaling a strategic shift in its diabetes treatment lineup. Healthcare providers are now advising patients on whether the oral version suits their treatment plans.
The injectable Ozempic, known generically as semaglutide, has faced supply shortages since 2022 due to high demand for both diabetes and weight loss. The oral tablet’s approval comes as competition intensifies in the GLP-1 drug market, with other companies developing similar alternatives. Pharmacies are expected to stock the new pill within weeks.
Patients switching from Rybelsus or injectable Ozempic should consult their doctors for dosing adjustments. The oral tablet’s smaller doses may require different titration schedules, and healthcare providers will monitor for side effects like nausea or digestive issues. Novo Nordisk has not announced pricing details for the new formulation.
Moving forward, the oral Ozempic could expand access to GLP-1 treatments for patients hesitant about injections. Doctors anticipate broader adoption if the pill proves as effective as the injectable version in real-world use. The shift also reflects growing demand for non-injectable diabetes medications amid the obesity treatment boom.
For now, the oral Ozempic tablet joins a crowded market of GLP-1 drugs, including Trulicity and Mounjaro, which offer similar benefits with varying administration methods.
What You Need to Know
- Source: Healthline
- Published: May 16, 2026 at 12:24 UTC
- Category: Health
- Topics: #health · #wellness · #medicine · #diabetes · #could-revolutionize-type · #diabetes-treatment
Read the Full Story
This is a curated summary. For the complete article, original data, quotes and full analysis:
All reporting rights belong to the respective author(s) at Healthline. GlobalBR News summarizes publicly available content to help readers discover the most relevant global news.
Curated by GlobalBR News · May 16, 2026
Related Articles
- Why ‘Ballmaxxing’ May Be More Dangerous Than You Think
- Weight Loss Surgeries Decline as GLP-1 Use Skyrockets. Why This Matters
- Olympian Allyson Felix on Why Rest and Recovery Beat Hustle Culture
🇧🇷 Resumo em Português
A busca por tratamentos mais práticos para a diabetes tipo 2 acaba de ganhar um novo capítulo nos Estados Unidos, com a aprovação de um comprimido oral do Ozempic, medicamento já conhecido por suas injeções. A novidade promete revolucionar o dia a dia de milhões de pacientes, oferecendo uma alternativa menos invasiva aos que dependem de aplicações diárias.
A aprovação da pílula pelo FDA (agência reguladora americana) chega em um momento crítico para o Brasil, onde mais de 16 milhões de pessoas convivem com a doença, segundo a Sociedade Brasileira de Diabetes. Especialistas brasileiros destacam que a chegada de versões orais de medicamentos como o Ozempic pode facilitar o acesso ao tratamento, especialmente para pacientes com dificuldades no uso de injeções ou que buscam maior comodidade. Além disso, a redução da dependência de aplicações diárias, como a do Rybelsus, já disponível no mercado, pode aumentar a adesão dos pacientes aos protocolos terapêuticos, melhorando o controle glicêmico e reduzindo complicações a longo prazo.
A expectativa agora é de que a Anvisa analise a liberação desse medicamento no Brasil, o que poderia transformar o tratamento da diabetes tipo 2 no país nos próximos anos.
🇪🇸 Resumen en Español
La Agencia de Alimentos y Medicamentos de EE.UU. (FDA) ha dado luz verde a la primera versión en comprimidos de Ozempic, un fármaco revolucionario en el tratamiento de la diabetes tipo 2 que promete simplificar la vida de miles de pacientes al eliminar la necesidad de inyecciones.
Esta aprobación marca un hito en la medicina, ya que hasta ahora los pacientes debían optar por inyecciones semanales o recurrir al medicamento oral Rybelsus, de administración diaria y menor adherencia. Los expertos destacan que la nueva pastilla de Ozempic podría mejorar la calidad de vida de los diabéticos al combinar la eficacia de su principio activo con la comodidad de un formato más accesible, aunque advierten sobre posibles efectos secundarios digestivos y la necesidad de evaluar su impacto real en la población hispanohablante, donde la diabetes tipo 2 tiene una prevalencia especialmente alta.
Healthline
Read full article at Healthline →This post is a curated summary. All rights belong to the original author(s) and Healthline.
Was this article helpful?


Discussion