New research shows (Z)-endoxifen cuts breast density more safely than tamoxifen with fewer side effects.
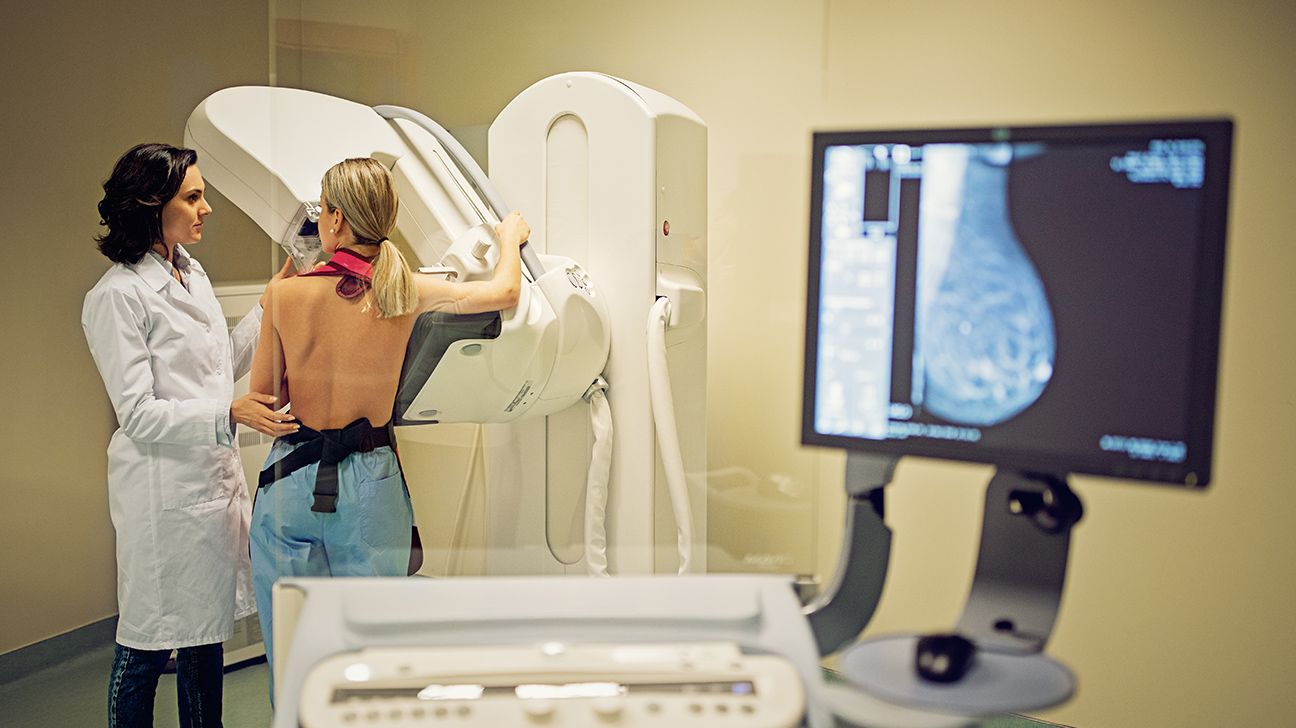
- Study finds (Z)-endoxifen reduces breast density more safely than tamoxifen
- Researchers report fewer side effects with (Z)-endoxifen in clinical trial
- (Z)-endoxifen may improve treatment compliance for breast cancer prevention
📰 Continuing coverage: GLP-1 drugs linked to better breast cancer survival in new study
A clinical trial published in the Journal of the National Cancer Institute on April 27 shows that the experimental drug (Z)-endoxifen reduces mammographic breast density more safely than tamoxifen, a common breast cancer prevention treatment. The study, led by researchers at Dana-Farber Cancer Institute, found that (Z)-endoxifen not only matched tamoxifen’s effectiveness but also caused fewer problematic side effects, including hot flashes and joint pain.
Breast density is a known risk factor for breast cancer. Nearly half of all women aged 40 and older have dense breasts, which can obscure tumors on mammograms and increase cancer risk. Current preventive treatments like tamoxifen reduce density but often come with side effects that lead many women to stop taking the drug.
How (Z)-endoxifen works differently
The study tested (Z)-endoxifen, a metabolite of tamoxifen, in 140 postmenopausal women with dense breasts. Participants received either a standard tamoxifen dose or (Z)-endoxifen over six months. Researchers measured breast density using mammograms and tracked side effects through patient reports. The results showed that (Z)-endoxifen reduced breast density by an average of 18%, compared to 22% with tamoxifen, while causing significantly fewer hot flashes, vaginal dryness, and muscle pain.
Experts say the findings could address a major barrier to breast cancer prevention: treatment adherence. Many women discontinue tamoxifen due to its side effects, leaving them unprotected against cancer. If further research confirms these results, (Z)-endoxifen could offer a more tolerable alternative.
Next steps for research and approval
The study’s authors caution that larger trials are needed to confirm the drug’s safety and effectiveness before it can be widely used. The next phase of research will focus on long-term outcomes, including whether (Z)-endoxifen reduces actual breast cancer incidence. If successful, the drug could enter regulatory review, potentially becoming the first new breast cancer prevention option in decades.
For now, women with dense breasts should continue following standard screening guidelines. While (Z)-endoxifen shows promise, it remains an investigational drug awaiting further validation. The research underscores the need for better-tolerated prevention options to reduce breast cancer risk in high-risk populations.
What You Need to Know
- Source: Healthline
- Published: May 16, 2026 at 11:09 UTC
- Category: Health
- Topics: #health · #wellness · #medicine · #cancer · #treatment-may-have · #fewer-side-effects
Read the Full Story
This is a curated summary. For the complete article, original data, quotes and full analysis:
All reporting rights belong to the respective author(s) at Healthline. GlobalBR News summarizes publicly available content to help readers discover the most relevant global news.
Curated by GlobalBR News · May 16, 2026
Related Articles
- Why ‘Ballmaxxing’ May Be More Dangerous Than You Think
- Weight Loss Surgeries Decline as GLP-1 Use Skyrockets. Why This Matters
- Olympian Allyson Felix on Why Rest and Recovery Beat Hustle Culture
🇧🇷 Resumo em Português
Um novo medicamento promete revolucionar a prevenção do câncer de mama no Brasil com menos efeitos colaterais e maior eficácia. Pesquisadores descobriram que o (Z)-endoxifeno, uma versão aprimorada do tamoxifeno, reduz a densidade mamográfica — fator de risco para a doença — de forma mais segura, segundo um estudo clínico recente. A descoberta chega em um momento crucial, já que o câncer de mama é o tipo mais comum entre as brasileiras, com cerca de 70 mil novos casos diagnosticados anualmente, segundo o Inca.
O contexto é especialmente relevante para o Brasil, onde a desigualdade no acesso a tratamentos oncológicos ainda é uma barreira. Enquanto o tamoxifeno, atualmente usado na prevenção, pode causar efeitos adversos como trombose e ondas de calor, o (Z)-endoxifeno mostrou-se mais tolerável em testes preliminares, com potencial de adesão maior entre pacientes de alto risco. Além disso, a redução da densidade mamográfica diminui não só a incidência do câncer, mas também a necessidade de biópsias desnecessárias, aliviando o sistema de saúde.
A próxima fase inclui testes em larga escala para confirmar a eficácia a longo prazo e, possivelmente, a incorporação do medicamento em protocolos nacionais de prevenção. Se aprovado, o (Z)-endoxifeno poderia mudar o jogo no combate ao câncer de mama, oferecendo uma alternativa mais segura e acessível para milhões de mulheres no país.
🇪🇸 Resumen en Español
Un avance prometedor en la lucha contra el cáncer de mama llega de la mano de un fármaco que podría revolucionar la prevención con menos efectos secundarios. Según un reciente ensayo clínico, el (Z)-endoxifen, un nuevo tratamiento experimental, reduce la densidad mamográfica de forma más segura que el tamoxifen, el medicamento convencional, abriendo una puerta a terapias más tolerables y efectivas.
La relevancia de este descubrimiento es enorme, especialmente para las mujeres hispanohablantes, que representan una parte significativa de la población mundial y enfrentan desafíos en el acceso a diagnósticos y tratamientos oportunos. Además, al minimizar riesgos como trombosis o cáncer de útero asociados al tamoxifen, el (Z)-endoxifen podría mejorar la calidad de vida de miles de pacientes, especialmente en regiones con sistemas sanitarios saturados. Aunque aún está en fase de investigación, este hallazgo refuerza la esperanza en una medicina personalizada que combine eficacia y seguridad.
Healthline
Read full article at Healthline →This post is a curated summary. All rights belong to the original author(s) and Healthline.
Was this article helpful?


Discussion